ENERGY TRANSFERS

Internal energy is stored by all macroscopic objects, which consist of a large number of molecules that generally have kinetic energy—possibly just from a jiggling motion—and exert electric forces on one another. Thus, there are potential energy makes up part of the internal energy.
Thermodynamics deals largely with the properties of macroscopic systems and in particular, energy exchanges between systems of different spatial regions of the same system. The objective of this essay is to outline the types of energy transfers that occur in thermodynamics, namely, work, heat and mass transport.
In Newtonian mechanics, in situations where two or more particles have kinetic energies and mutual gravitational and/or electric potential energies, a great principle of physics emerges: The conservation of mechanical energy. What this means is that the sum of all the kinetic and potential energies is a non-changing constant. There can be a conversion of kinetic to potential energy or vice versa but the total remains unchanged. Symbolically, using KE, PE, and E for kinetic, potential and total energy,
Total KE + Total PE = E = constant
Here, all objects are idealized either as point particles or so-called rigid bodies. In either case there is no such thing as internal energy; i.e., neither point particles nor rigid bodies can store energy within them. Point particles, by definition, don't even have an interior, and rigid bodies are assumed to be made of rigidly connected "molecules" that cannot jiggle. In physics language, there are no internal degrees of freedom in either case.
All experimental evidence shows that energy is conserved also for thermodynamic processes. In this case, where macroscopic matter is involved, there are so-called internal degrees of freedom. That is, each molecule in a thermodynamic system can jiggle and can also rotate and vibrate relative to its center of mass. Each such way to store energy is associated with degrees of freedom. Jiggling in three-dimensional space has 3 degrees of freedom (the x, y, and z directions); rotation and vibration have 2 degrees and 1 degree of freedom, respectively. The total energy stored by all molecules can be altered, thereby altering the thermodynamic system's internal energy, and also the internal energy of the surroundings. The thermodynamic system of interest plus its surroundings form an isolated system, for which the total energy is conserved.
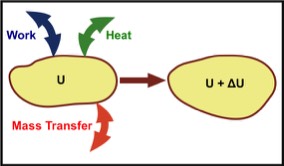
The internal energy of a thermodynamic system, typically denoted by the symbol U. For a given thermodynamic system, it is the average of the sum of molecular kinetic and potential energies. I use the term average because no real system is isolated from its environment and thus relatively small additions and subtractions to U typically occur.
The quantity that is of importance for thermodynamic processes is the internal energy change ∆U. It is generally postulated in thermodynamics that U exists for each equilibrium thermodynamic state. That is, U is a meaningful quantity whose changes can be measured as explained here. The value of U itself is typically not of interest. However, it is assumed that U depends only on the macroscopic state it is in.
The thermodynamic state can be specified by a small number of parameters such as volume, total mass, and temperature. For this reason, internal energy is often called a state function.
• First law of thermodynamics
If a thermodynamic system is heated such that its volume does not change, then zero work is done on it by the surrounding atmosphere. The corresponding internal energy change is ∆U = Q, where Q is the amount of energy added by heating. The same expression holds for cooling of the system, but with Q being negative rather than positive. This is nothing more than a statement of conservation of energy.
Another way to modify the energy of a thermodynamic system is via physical work—namely, exerting a force on it that acts through a non-zero distance. An example is bending a wire coat hanger back and forth repeatedly, in which case its temperature rises. Here the system is the wire and the surroundings include you, the bender and the surrounding air. Such bending generates a temperature change, but this not a heat process. It is the work of bending the wire that increases the coat hangers internal energy and temperature. Because the wire becomes warmer than its environment it will then release energy by a heat process that heats the surrounding air. In this case the energy conservation equation is
∆U = Q + W
This equation is commonly referred to as the first law of thermodynamics (FLT), a generalization of the energy conservation principle of Newtonian mechanics. I define W to be the positive work done ON the system by the person doing the bending; and Q to be the energy transfer to the metal. In the envisaged case, Q < 0, because energy is sent to the environment via a heat process.
If the released energy equals the work of bending, then -Q = W and ∆U = 0. However, it is likely that in actuality Q + W ≠ 0; i.e., the magnitude of the released energy does not equal the work done, leaving the bent wire with an altered internal energy. This is expected because bending modifies distances between some molecules in the metal—increasing some potential energies and decreasing others as adjacent molecules become more or less separated.
In the latter example, a work process will induce a heat process because the wire becomes warmer than the surrounding air. In contrast, if we heat a steel solid, it will expand a bit, thereby pushing some of the surrounding air away; i.e., the solid does positive work on the atmosphere; i.e., W < 0, while Q > 0. An equivalent view is that the atmosphere does negative work on the solid. In a nutshell, work processes can induce heat processes and vice versa.
Work is well defined in mechanics as force times displacement. Thus a pure work process with Q = 0—called an adiabatic process—can be used to determine ∆U; i.e., ∆U = Wadiabatic. Once this is done, then for another process that connects the same two states, and for which Q ≠ 0, we have Q = ∆U - W, or
Q = Wadiabatic - W
This somewhat subtle argument shows how heat Q can be determined purely from two work measurements.
• Generalization and More Examples
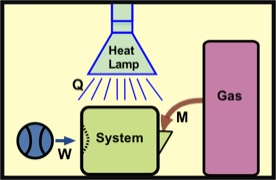
∆U = Q + W + M,
Here, M connotes "mass transfer energy." For most of the examples I'll examine, M is either zero or is negligible.
Note that any chosen thermodynamic system must have a well-defined boundary. Otherwise the distinction between system and surroundings would be unclear. For example, the air in your room is a thermodynamic system whose walls, floor, and ceiling form the system boundary.
In a typical house, heat processes occur through the walls, with Qwalls > 0 in summer and Qwalls < 0 in winter. Also, air flows through small openings around doors and windows, and both M > 0 or M < 0 are possible. In winter, an energy input might be provided by a furnace burning natural gas with Qfurn > 0, and cooling might be accomplished by an air conditioner, with Qcool < 0.
If the system of interest is the air inside a balloon, the inner surface of the balloon’s rubber skin defines the boundary. But if the system is taken to be the air plus balloon skin, then the outer surface of the balloon’s skin defines the boundary. In cases where the boundary’s surface area and shape, and perhaps its volume, can change, we say the boundary deforms.
An interesting example is what happens energetically when you arise from a seated position. As you stand up, many of the molecules in your body are lifted upward. Perhaps surprisingly, the only external work done on you is by the downward gravitational force, and that work is negative(!), because the gravitational force acts downward and the displacement of each molecule that moves is upward. During this process, the mutual gravitational potential energy for you and Earth increases.
- Example: Rising from a seated position
The energy that makes rising from a seated position possible comes from the digestion of food, which releases energy that fuels your muscles. This energy from your digestion is sometimes referred to as coming from food calories. This is an unfortunate misnomer because the energy is not really stored by foods per se. Rather, digestion is a consequence of chemical reactions of carbohydrates, proteins, and fats with oxygen from the air. If your body is taken to be the thermodynamic system, the term M is not zero, but includes energy taken in via oxygen and that breathed out as carbon dioxide. The point is that food alone cannot provide energy to you. Combining it with oxygen is essential.
The energy released in digestion and in fueling your muscles goes into increasing your gravitational potential energy as you stand up. You also heat your cooler surroundings as your body maintains normal body temperature of about 37 °C = 310 K, while powering your heart, kidneys, and the like. Such heating is why concert halls filled with people become unbearably warm unless cooler air is pumped in.
The external work done on the body is negative (W < 0), as the body itself does positive internal (not external) work associated with your muscles doing the lifting. Notably, the upward external force of the floor on your feet as you stand does zero work because it acts through zero displacement. Although this "floor force" is essential because without it, you could not stand up, it does zero work on you. If you find this counter-intuitive, it is!
Although not of primary interest here, the upward force from the floor is, and must be, greater than your body weight as you raise your body from sitting to standing. This enables you to change the momentum associated with the movement of your center of mass. A similar phenomenon occurs if you put on roller skates and push away from a wall. The wall's force does zero work (which is also counter-intuitive), but enables your momentum to increase from zero to a non-zero value as you push against the wall.
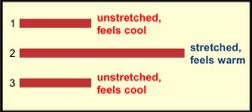
A really nice, remarkably simple, example is to take a rubber band, hold it in front of you in a taut but not-stretched configuration, and touch it to your forehead briefly. Typically, it feels a bit cool because it is at room temperature—about 22 °C, which is cooler than your body's 37 °C. This taut position is labeled 1 at the left. Now quickly stretch the rubber band and immediately touch it to your forehead (position 2) briefly. Does it feel warmer? Most rubber bands give this effect.
This is a simple activity to try yourself. The quick stretch makes the rubber band feel warm, just as "working" a metal coat hanger makes it feel warm. The stretching is a work process—work by you on the rubber band. This increases the internal energy of the rubber band in two ways. Long-chain rubber molecules that were largely coiled are now stretched, which increases their potential energy, and the quick stretch also causes molecules to jiggle more than they were initially; i.e., higher kinetic energy. Both effects increase the internal energy.
On the other hand, if you stretch the rubber band quickly and immediately let the rubber band pull your hands closer together (to position 3), and touch it to your forehead, it should feel a cooler, as it did initially. The rubber band did positive work on you (and you did negative work on it), as it returns to the original configuration.
If you quickly stretch a rubber band and keep it stretched in position 2 for 10 seconds, it will have time to transfer energy to the lower-temperature surrounding air via a heat process, cooling to room temperature—even though still stretched. Check this out. What did you feel? Do you understand why?
In a later essay, I'll discuss the interesting entropy behavior for stretching and un-stretching, and its remarkable and perhaps surprising connection to a technique for achieving ultra-low (cryogenic) temperatures.
• Heat Process Types
By definition, heat processes are energy transfers that are induced by temperature differences. Notably, such energy flows are from higher to lower temperatures. I've illustrated them with some examples. Other examples are heating hot water for tea using a high-temperature electric burner, heating a room with a central natural gas-fueled forced air heating system, and radiation energy from the hotter sun heating the cooler earth. There are three basic types of heat processes: conduction, convection, and radiation. I outline each briefly in what follows.
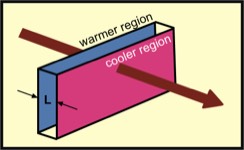
Energy transfers by conduction occur within matter when there is a temperature gradient; i.e., the temperature varies from one spatial region to another. This is illustrated in the figure at the right, which shows a slab of material that is kept at a higher temperature on on side (blue) and a lower temperature at the other (pink). The slab has thickness denoted by L. For example, the slab might be 5 cm thick; i.e., L = 5 cm. For the example of heating water in a pot, the heat source is the electric burner and the thickness L is that of the pot's bottom. Energy flows from the burner, through the pot, to the water.
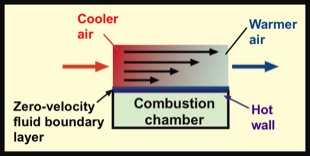
Energy transfer by convection occurs when a moving fluid (liquid or gas) travels from a region at one temperature to another region at a different temperature. In essence, the fluid is a vehicle that picks up energy in one region and and gives up energy in another, or vice versa.
For the example of forced-air heating, cool air enters the heating system and flows past a hot wall of a furnace, which warms it. The warm air is delivered to the space being heated.
Another familiar example is heating by a portable electric hair dryer. Air is passed over an electric heating element, which heats it. The resulting hot air can then be used to speed up hair drying.
In the case of cooling by air conditioning, air passes over a cold coil through which a cold refrigerant flows, dumping energy via conduction and convection. This cools the air, which is then sent into the space being cooled.
- Radiation

We understand "heat" radiation energy as being carried primarily by the infrared (IR) part of the electromagnetic (EM) spectrum. The full EM spectrum includes phenomena from‚in order of increasing frequency—AM radio, FM radio and TV, microwaves, infrared, visible light, ultraviolet, x-rays and finally, gamma rays. Infrared radiation is similar to light radiation, except that its frequencies are much smaller. Our bodies are able to detect infrared radiation and when it does, it sends a message to our brains that there is something warming us. Standing in front of a blazing fireplace is a familiar example.
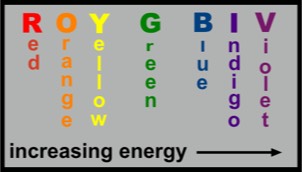
A typical candle flame is about 1,000 °C and is yellowish. In contrast, when natural gas burns in air the flame color is blue and the temperature is nearly 2,000 °C. This supports our understanding that the color spectrum from red to violet represents photons of light of increasingly higher energy. This behavior is summarized in the figure at the left.
For an incandescent light bulb, an electric current source provides external work to generate the current. As the electric current flows through the tungsten filament of the lamp, it transfers energy to the metal's molecules, which become excited, inducing a release of IR radiation. This is another example of a process that entails both work and heat, and of course, a change of internal energy.
Photon frequencies just smaller than IR radiation are microwaves. Modern microwave ovens operate generate such radiation to heat food. The IR radiation works largely by heating the water in foods. Specifically it makes H2O molecules that absorb its photons rotate. This is a work process that generates rotational kinetic energy. Ultimately, this energy becomes more jiggling and higher temperatures of the water molecules.
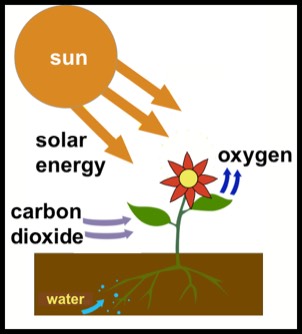
Wikipedia's entry for photosynthesis defines it as "a process used by plants and other organisms to convert light energy, normally from the sun, into chemical energy that can be used to fuel the organisms' activities." This statement, though common and pleasing, is not entirely accurate. More correctly, the energy released when foods are digested comes not strictly from the plant material, but by a chemical reaction that combines the plant material with oxygen from the atmosphere. We may envisage the energy to be stored by the combination of plant material and oxygen.
An over simplified version of the photosynthetic process envisages water and carbon dioxide combining under the action of sunlight to form glucose, which is a sugar in the family of carbohydrates. The process is illustrated at the right. The chemical reaction is:
6 H2O + 6 C02 + solar energy → C6H12O6 + 6 O2
Similarly, the following reaction, called combustion, is
C6H12O6 + 6 O2 → 6 H2O + 6 C02 + heating energy
In the photosynthetic process, energy is added to the water and carbon dioxide molecules to form glucose plus oxygen. Thus, there is more energy stored by the molecules on the right than on the left. In the combustion process, the "extra" energy of the glucose plus oxygen becomes energy of heating.
The chemical reaction formulas for photosynthesis and combustion might give the impression that each process is reversible, giving the other. However, in the thermodynamic sense, this is not so. Photosynthesis requires a high-temperature source like the sun to radiate energy to the cooler earth. This is not the reverse of combustion, which requires a high-temperature spark to initiate the combustion process (although a more complex version occurs regularly in the human body, at about 37 °C), ultimately sending energy to the cooler surrounding air. Indeed, both photosynthesis and combustion are irreversible thermodynamic processes. I'll have more to say about this in future essays.